Patient-Based Therapeutics
AcuraStem is a patient-based biotechnology company pioneering how treatments are developed for neurodegenerative diseases — including sporadic ALS and FTD — using our proprietary, best-in-class, disease-modeling platform, iNeuroRx®, the gold standard for discovering novel, effective and broadly-acting treatments.
Patient Focused




learn about our work
Neurodegenerative Disease Areas


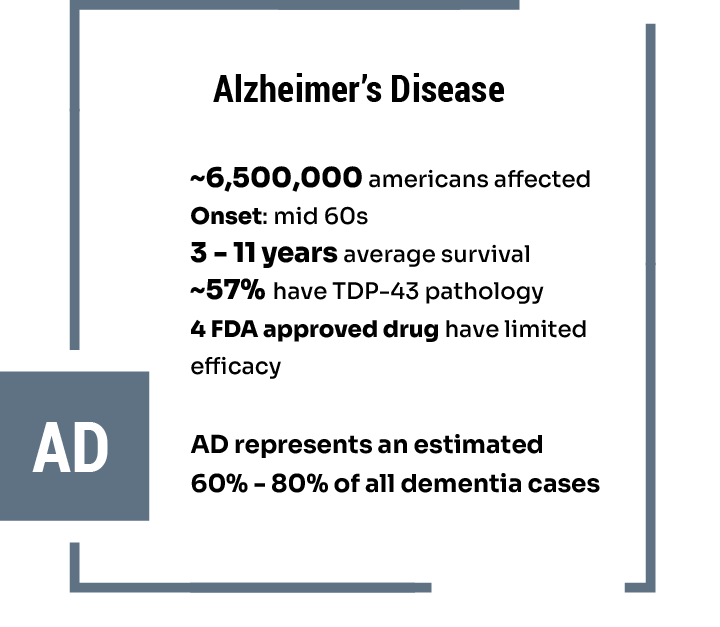
Latest News
AcuraStem, a patient-based biotechnology company advancing treatments for amyotrophic lateral sclerosis (ALS), frontotemporal dementia (FTD), and other neurodegenerative diseases, announced today that CEO Sam Alworth has been invited to speak at the RNA Leaders USA Congress, scheduled for September 4-5, 2024, at the San Diego Convention Center.
AcuraStem, a patient-based biotechnology company pioneering the development of treatments for neurodegenerative diseases, today announced the appointment of Dr. Ananya Sengupta as Associate Director of Program Management
AcuraStem’s Associate Vice President, Discovery and Early Development, Dr. Wen-Hsuan (Wen) Chang, will present at the 5th International Oligonucleotides and Peptides Conference (IOPC) on June 26, 2024, in Milan
